New research into the reasons that lithium-ion plating occurs on batteries could open the door to faster electric vehicle charging, extended range, and longer life spans for batteries.
Lithium ion plating has emerged as a problem for EV batteries because it can affect performance, slow acceleration, impact fast charging speeds and, in some cases, pose an increased fire and safety risk.
A new study, published in Nature, suggests a solution by tweaking the microstructure of a graphite anode, and changing how a battery is charged, could reduce the amount of lithium ion plating that takes place over time.
“Assisted by a pioneering 3D battery model, we can capture when and where lithium plating initiates and how fast it grows,” says study leader Dr. Xuekun Lu from Queen Mary University of London.
“This is a significant breakthrough that could have a major impact on the future of electric vehicles.”
What is lithium plating
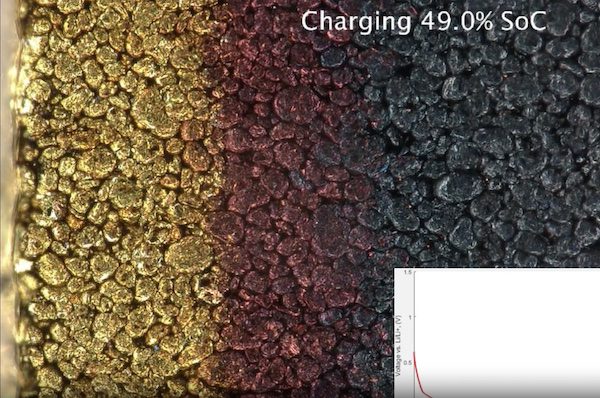
Lithium plating is when lithium ions build up on some parts of the surface of the anode.
It is generally caused by charging at low temperatures or using high currents – a requirement of fast charging infrastructure – when the surface of the anode is temporarily saturated with lithium ions. Instead of ‘intercalating”, or entering the anode structure, the ions build up on the outside as a metallic layer.
Over time small build ups might be removed in later discharges, but other times the new layer will become self-reinforcing.
Once this starts, it consumes lithium that could be used in the battery, reduces the porosity of the anode for other lithium ions to enter, and reduces the area on the anode that can react.
What that means is reduced battery performance now, to the point where it can slow an electric vehicle’s (EV) acceleration ability, and a shorter lifespan for the unit. In some cases, it can cause short circuits and fires.
Fixing the plating problem
The research into how to fix this, using high-fidelity 3D microstructural-resolved phase-field modelling, was undertaken by a collaboration of UK and US researchers hailing from a swathe of storied institutions – think MIT, the Faraday Institution, Oxford and the Beijing Institute of Technology, among others.
What they found is that the particles that make up the anode are not uniform and therefore make it more or less likely for lithium ions to easily intercalate, or not.
Manipulating the particles of the anode so they’re more uniform instead of being randomly distributed would allow for a more homogeneous reaction between the lithium ions and the anode and prevent the build up of a lithium layer in some areas but not others, Lu says.
However, this is also quite challenging.
Another, simpler, fix would be changing how fast chargers work.
The study found constant-current voltage is not suitable for fast charging because of the high current that needs to be used.
Already a range of different options have been canvassed by researchers elsewhere, such as pulsed current, varied current, multistage constant current, or a hybrid profile combining different modes.
The study authors say for a very fast full charge in 20 minutes, there should be a rest period for 3 minutes at 45 percent state of charge (SOC) for an electrode with a real capacity up to 2 mAh cm−2 (discharge capacity per unit area).
For a 3 mAh cm−2 electrode, the optimal relaxation time is at 45 per cent SOC for a 20 minute charge, 30 per cent SOC for a charge that takes a slower 30 minutes, and 20 per cent SOC for a charge that takes 60 minutes.
By giving the battery time to “relax”, it gives the anode time to absorb the lithium ions, helps with recovery by stripping away any plated lithium that is reversible, and reduces capacity loss in-the-moment by stopping the formation of so-called “dead lithium”.

Rachel Williamson is a science and business journalist, who focuses on climate change-related health and environmental issues.