Emissions from passenger and light commercial road vehicles make up 10 per cent of Australia’s total CO2emissions, so reducing these emissions can make a significant contribution to reducing Australia’s CO2 emissions.
The purpose of switching from fossil fuels (petrol/diesel/LPG) to low-emission alternative fuels is to reduce/mitigate climate change by reducing total CO2emissions.
If an alternative fuel/powertrain can be deployed which has a lower cost per km than fossil fuels, is fit-for-purpose, and has no significant disadvantages (e.g. power, torque, safety), then the market will probably naturally and speedily gravitate to the new fuel/drivetrain.
Thus, any viable market-changing solution must reduce costs, not just emissions.
If the alternative fuel is produced using renewable energy, then transport operational emissions can be eliminated.
If the fuel is produced using dirty energy sources which produce CO2 emissions during production, then the CO2-emissions / climate change mitigation objective has not been achieved; all that has happened is that emissions have been moved somewhere else, i.e. from tailpipe to the sources of the fuel.
In fact, emissions could even be worse.
This series of two articles examines hydrogen to see how it stacks up against these criteria, viz, lower emissions and lower cost. (The second part can now be found here).
Note: A light vehicle, such as an average-sized car (e.g. Toyota’s Mirai Hydrogen Vehicle, similar in size to a Camry), will travel up to 100 km on a kilogram of Hydrogen. A Mirai has a 5kg tank. Larger vehicles will obviously travel smaller distances on the same 1 kg of Hydrogen.
Production materials/feedstock
Producing Hydrogen requires chemical processing of a feedstock material. The feedstock options are water, methane gas, or coal. Firstly, let’s examine each production option for energy intensity, emissions intensity and other inputs:
Electrolysis (using electricity & water)
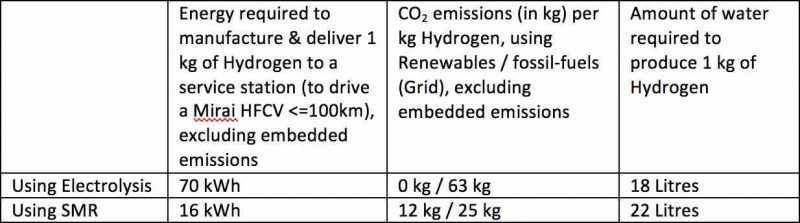
To produce Hydrogen from water, a chemical process called electrolysis is used. This process is highly energy intensive and requires about 55 kWh of energy to produce 1 kg of Hydrogen. 55 kWh is enough energy to power the average Australian household for 3.5 days.
The amount of water required is also significant – about 18 litres of water per kg of Hydrogen (=100km for a Mirai). In a country which will increasingly be affected by longer, drier, more frequent droughts, due to climate change, the supply of water for this process could become very unpopular, not to mention a risk.
If renewable energy is used to perform electrolysis, then the process is carbon neutral. However, if the process is powered by the standard mix of generators in the local grid, then emissions remain high.
Using the current average Australian grid energy intensity of 0.9kg CO2-e/kWh, the emissions associated with producing 1 kg of Hydrogen would be 50 kg CO2-e.
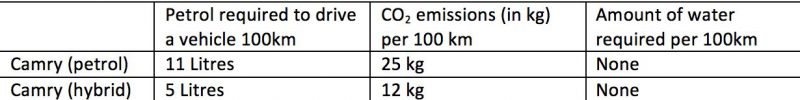
In contrast, a petrol Camry, driven in urban conditions, produces around 25kg of CO2-e per 100 km.
Thus, this production process uses substantial quantities of energy, is consequently expensive, and, unless it is powered exclusively by renewable energy, will also produce more emissions than a petrol vehicle.
Steam Methane Reforming (using methane & water)
Hydrogen can also be produced from methane using established industrial processes, i.e. Steam Methane Reforming (“SMR”), but unfortunately CO2is produced as a by-product and water is also consumed (‘steam…’).
Specifically, manufacturing Hydrogen from methane gas produces 12kg of CO2and uses 22 Litres of water for every 1kg of Hydrogen gas.
Thus, producing Hydrogen from methane produces the very emissions that the process is trying to eliminate, and consumes water.
Methane gas is typically used to power the process, requiring an additional 1kWh of methane gas to be burned for production of the steam.
Unfortunately, the financial cost of producing Hydrogen using methane gas is lower than using electrolysis, and so it is hard to see how a free market would buy hydrogen from any source other than methane, thus locking in CO2emissions at the refinery-end, instead of at the tailpipe, and defeating the purpose of the exercise.
Further, due to the shortage of methane in Australia, (and water, as described in the previous section), relying on methane is also a risk, both from an availability perspective as well as from a supply & demand-driven cost point of view.
The buyer of Hydrogen for their vehicle is unlikely to win from suppliers having this level of market-control.
Coal Gasification (using coal and water)
Coal can be converted into Hydrogen via expensive, energy-intensive, water-intensive industrial processes but the process also produces CO2, which again defeats the primary purpose of switching to Hydrogen – i.e. the reduction of CO2 emissions. Coal will therefore not achieve the objective of reducing costs and emissions per km.
Despite this, this production method is worth mentioning, given the Japanese funding provided for the La Trobe Valley pilot project.
The Japanese government has announced that they have no interest in emissions reduction until 2030-2040; the sole focus is on switching the country to a Hydrogen economy (for security reasons), regardless of cost.
The La Trobe pilot project, which is quite small and funded by the Japanese government, will use 160 tonnes of brown coal, produce 100 tonnes of CO2 and produce just three tonnes of Hydrogen.
Distribution
Once Hydrogen has been produced at a centralised industrial refinery – whether by electrolysis or SMR – it will need to be transported to a service station for retailing.
In order to make the gas economic to transport, it will need to be compressed – the usual pressure is 700 bar (equivalent to 700 times atmospheric pressure).
Alternatively, it can be liquified which requires less space but requires even more pressure and extremely low temperatures (-250C) – which require even more energy.
Containers to hold the Hydrogen need not only to be strong enough to withstand pressures of 700 bar while being transported by truck, and safe enough to tolerate accidents, but they also need to be made of specialised, expensive materials which will retain the Hydrogen, as Hydrogen will rapidly leak out of conventional materials.
At the service station, Hydrogen storage tanks also need to be constructed out of the same strong, safe, leakproof materials.
An alternative to distributing Hydrogen as a compressed gas or as a liquid is to convert Hydrogen into another chemical, for example, ammonia.
To perform this conversion uses still more energy and further increases costs. While transport of ammonia by truck is comparatively simple, the ammonia would need to converted back to Hydrogen at the service station and the Hydrogen compressed to 700 bar.
This is complex, expensive and may not be appropriate/possible at a service station in a suburban setting. Ammonia would thus further add to costs and emissions.
The amount of energy budgeted for the distribution phase, using compression, is usually calculated at 15 kWh per kg of Hydrogen.
If this energy is produced from renewable energy, there are no CO2emissions; however, if compression is performed using the average Australian grid emissions for example, then a further 13.5 kg of CO2 emissions are generated.
These figures do not include embedded emissions from producing and maintaining either the storage & transport assets, or the refineries – and does not include the liquefaction or ammonia options.
In summary:
For comparison:
Figures are based on urban consumption
The Distribution of electricity
The grid should also be involved in any examination of distribution.
The grid will be needed to deliver the electricity required to power these production facilities, and the use of compression or liquefaction or ammonia-as-a-carrier for logistics during distribution to service stations.
Pumps at service stations will also need power to re-compress storage tanks as Hydrogen is pumped out.
The energy costs of switching the Australian fleet to Hydrogen have been calculated at around 150 TWh – over 60% of all electricity currently generated by today’s Australian grid.
Since the grid has not got a spare 60% of capacity available – especially in summer – that means a massive expansion of power stations (generation) and the electricity transmission grid (poles & wires) would be required. This approach would also be a substantial threat to grid stability.
Such a grid expansion would therefore be extremely expensive.
In contrast, one might ask what the effect a switch to Lithium Battery Vehicles might have on the grid instead of a switch to Hydrogen Fuel Cell Vehicles.
The effect is quite small, requiring just 15% of the current production of electricity. Some electricity would potentially be used more at night (due to lower prices for electricity at night and because many people will plug in when they go home at night) in order to top-up a vehicle daily.
As the grid has excess capacity overnight, this outcome is actually good for the grid.
In addition, some drivers might just top-up once a week on the weekend if they have solar on the roof, thus largely relieving the grid of responsibility for keeping them mobile.
In the next article, costs of Hydrogen vehicles and infrastructure are examined.

Bede Doherty is an independent consultant in climate change mitigation, specialising in transport including fuels/powertrains, fuelling infrastructure and emissions.