New research has demonstrated the cause behind the mysterious short circuiting and failure of lithium metal batteries boasting solid electrolytes, a discovery that could lead to new designs and battery production methods that help to avoid the problem.
A team from Stanford University and the US Department of Energy’s SLAC National Accelerator Laboratory has demonstrated through more than 60 experiments that the ceramic electrolytes at the heart of many of today’s batteries are often littered with nanoscopic cracks and fissures.
Many of these imperfections in the ceramic electrolytes can measure as little as 20 nanometres wide – as compared to a sheet of paper, which measures around 100,000 nanometres thick.
But due to mechanical stresses like bending or twisting, fissures and cracks can be created that allow the lithium to intrude, causing the battery to short circuit or fail.
“Just modest indentation, bending or twisting of the batteries can cause nanoscopic fissures in the materials to open and lithium to intrude into the solid electrolyte causing it to short circuit,” says William Chueh, senior author and an associate professor of materials science and engineering in the School of Engineering, and of energy sciences and engineering in the new Stanford Doerr School of Sustainability.
“Even dust or other impurities introduced in manufacturing can generate enough stress to cause failure.”
The new research serves to explain through rigorous and statistically significant experiments how nanoscale defects and mechanical stress are causing solid electrolytes to fail.
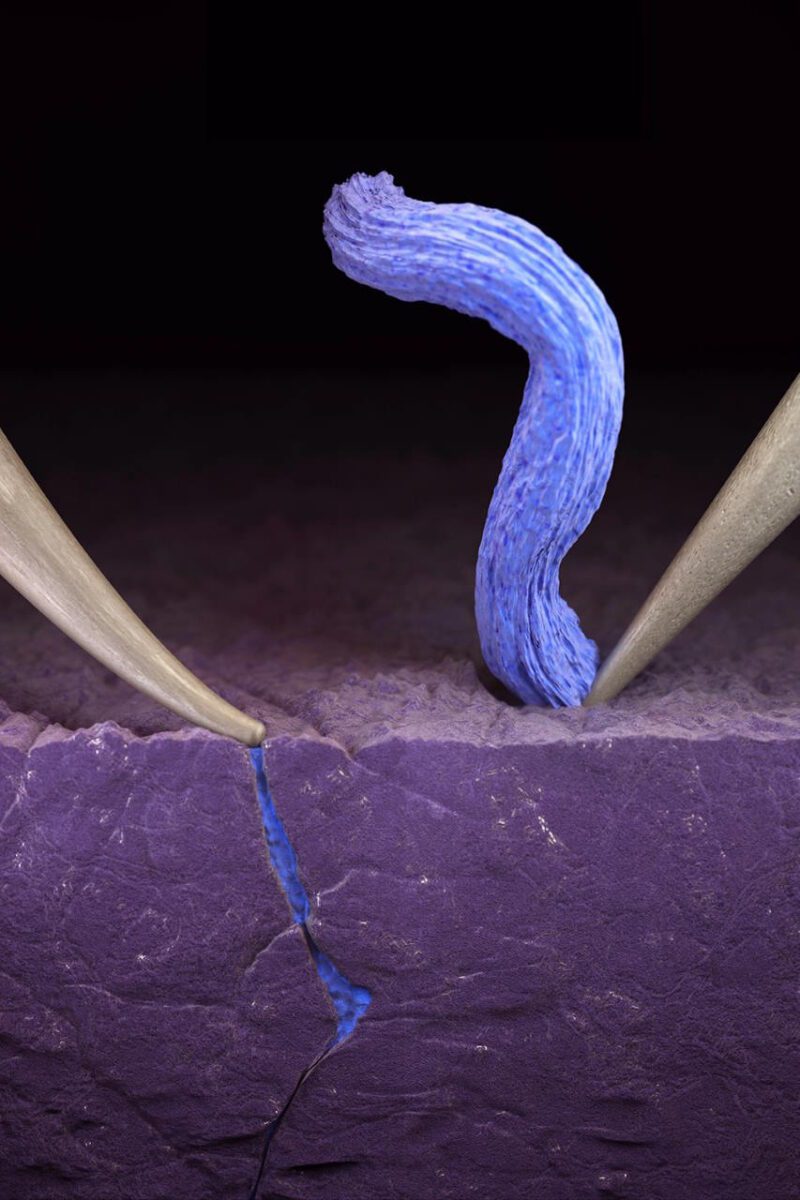
Each experiment conducted by the researchers applied an electrical probe to a solid electrolyte, creating a miniature battery, and used an electron microscope to observe fast charging in real time.
Next, the researchers used an ion beam as an ultra-precise scalpel to determine why the lithium collects and pools on the surface of the ceramic electrolyte in some places, but burrows deeper and deeper in other locations – eventually causing the lithium to bridge across the solid electrolyte, creating a short circuit.
After all the experiments were done, the researchers concluded that the difference was pressure.
Specifically, when one of their electrical probes merely rests on the surface of the electrolyte, the lithium gathers across the top of the electrolyte, even when the battery is charged in less than one minute.
When the probe presses into the ceramic electrolyte, however – mimicking the mechanical stresses of indentation, bending, twisting, etc – the researchers determined that it was more probable that the battery short circuits.
“Given the opportunity to burrow into the electrolyte, the lithium will eventually snake its way through, connecting the cathode and anode,” says Geoff McConohy, a co-lead author of the study.
“When that happens, the battery fails.”
Next up for the researchers is using their new knowledge to create methods to use these same mechanical forces intentionally so as to toughen the material during manufacturing. The researchers are also planning on looking at ways to coat the electrolyte surface to prevent cracks, or to repair cracks if they emerge.
Joshua S. Hill is a Melbourne-based journalist who has been writing about climate change, clean technology, and electric vehicles for over 15 years. He has been reporting on electric vehicles and clean technologies for Renew Economy and The Driven since 2012. His preferred mode of transport is his feet.